CBS News Live
CBS News Texas: Local News, Weather & More
Watch CBS News





Prayers are vehicles for answers, but a group that came to a church on Dallas' west side looking for solutions to gun violence after a school shooting left with none.

Officers determined the man had injuries "on his hands consistent with the incident," police said Tuesday.

Dallas megachurch pastor Dr. Frederick Haynes III will step down as president and CEO of Rainbow PUSH Coalition.

Sadness and questions still linger on Lime Leaf Lane.

We'll see another round of potentially strong thunderstorms in parts of North Texas Thursday.

The 17-year-old accused of shooting a fellow student at Wilmer-Hutchins High School on Friday morning "intentionally and knowingly" brought a revolver to school, the arrest affidavit states.

More than 300 drugs are in short supply, according to the American Society of Health System Pharmacists.

A Billy Joel special on CBS and Paramount+ will air again after it was cut off in the middle of the singer's performance of "Piano Man."

The $872 million most likely excludes any amount UnitedHealth may have paid to hackers in ransom.

Duncanville Animal Control took possession of the dogs.

Tyrese Simmons, who was charged with murder in the killing of Brandoniya Bennett, will plead guilty to manslaughter.

Raynaldo Ortiz's motive has never been perfectly clear, but prosecutors believe he was angry he was being investigated for errors in his own surgeries.

The City of North Richland Hills says drone flights could begin as early as Monday of next week. The drone deliveries are a partnership between Walmart and a company called Wing and will be offered in North Richland Hills and surrounding cities. Other cities in North Texas, like Rrisco and Little Elm, already offer similar drone delivery.
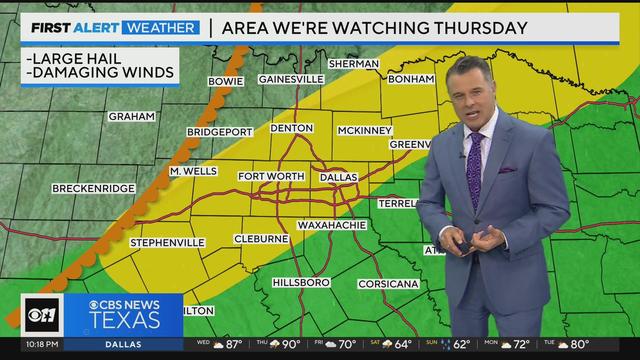
Temperature will approach 90 degrees Thursday before a cold front passes through. Strong to severe storms are possible.

Almost every day, someone is killed in the U.S. during a high-speed police chase.
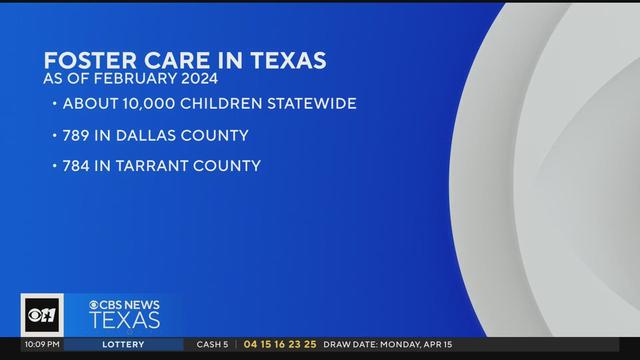
It took just one day for Texas to appeal a federal judge's ruling concerning the state's foster care system. A federal judge is fining the state $100,000 a day for contempt and neglecting to adequately investigate allegations of abuse against some who are in the system. As of February, there are about 10,000 children in the system statewide. An estimated 789 boys and girls are in foster care in Dallas County, and 784 in Tarrant County as of April 16, 2024.

Following Frederick Haynes' resignation, Rev. Jesse Jackson said his son, Yusef Jackson will continue to serve as Chief Operating Officer.

Temperature will approach 90 degrees Thursday before a cold front passes through. Strong to severe storms are possible.

We'll see another round of potentially strong thunderstorms in parts of North Texas Thursday.
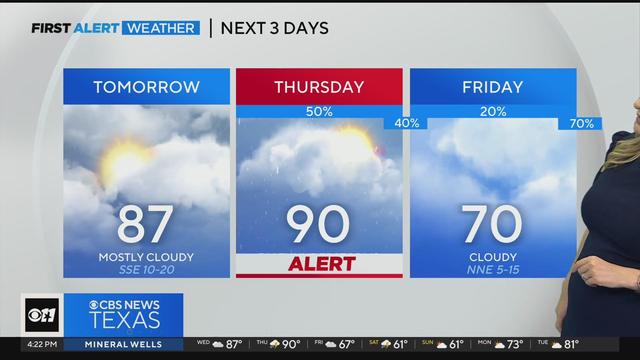
Wednesday, we'll see mostly cloudy skies with highs in the mid to upper 80s. Highs will be near 90 degrees on Thursday! We'll also see another round of potentially strong thunderstorms in parts of North Texas this Thursday.

Texas police departments have the discretion to determine the frequency and extent of additional driving training for their officers. While some require driving training yearly or every other year, others do not.

Some departments opt to melt the firearms down, while others choose to crush them. However, there are instances where firearms, or at least parts of them, escape destruction altogether.

Several police departments told the CBS News Texas I-Team they were unaware of this practice, even though it was stated in the contracts they signed with the company, Gulf Coast GunBusters.

It's a complicated process that not everyone qualifies for.

Tuesday, federal prosecutors called their first witnesses against Dr. Raynaldo Ortiz.

Michael Lorenzen pitched five spotless innings in his Texas debut and three relievers completed a five-hit shutout as the Rangers topped the Detroit Tigers 1-0.

Caitlin Clark has been selected with the No. 1 pick in the WNBA draft by the Indiana Fever.

"Scottie's a very humble guy. Wonderful personality and a great representative for Highland Park and for Dallas."

World no. 1 Scottie Scheffler finished 11 under to win his second Masters Tournament in three years Sunday.

Here's how much the winner took home last year, and what other golfers made.

Prayers are vehicles for answers, but a group that came to a church on Dallas' west side looking for solutions to gun violence after a school shooting left with none.

Officers determined the man had injuries "on his hands consistent with the incident," police said Tuesday.

Dallas megachurch pastor Dr. Frederick Haynes III will step down as president and CEO of Rainbow PUSH Coalition.

Sadness and questions still linger on Lime Leaf Lane.

We'll see another round of potentially strong thunderstorms in parts of North Texas Thursday.

Texas police departments have the discretion to determine the frequency and extent of additional driving training for their officers. While some require driving training yearly or every other year, others do not.

Some departments opt to melt the firearms down, while others choose to crush them. However, there are instances where firearms, or at least parts of them, escape destruction altogether.

Several police departments told the CBS News Texas I-Team they were unaware of this practice, even though it was stated in the contracts they signed with the company, Gulf Coast GunBusters.

It's a complicated process that not everyone qualifies for.

Tuesday, federal prosecutors called their first witnesses against Dr. Raynaldo Ortiz.

Dallas' mayor is also calling for city council members to agree not to include a golden parachute clause in the next city manager's contract.

The Senate is tasked with the trial after the House impeached Mayorkas earlier this year. Senate Democrats are expected to move to quickly quash the effort.

Seven Manhattan residents were selected Tuesday afternoon to serve on the jury in former President Donald Trump's criminal trial.

If approved by voters during the May 4 municipal election in Frisco, Prop A would institute civil service protections for firefighters. Prop B would provide collective bargaining over pay, benefits, and workplace conditions between firefighters and the city. Bill Woodard, a Frisco City Council Member since 2016, opposes both propositions.

Frisco Firefighters Association President Matthew Sapp told CBS News Texas after city leaders rejected using meet and confer to discuss their pay and benefits, their members decided to go directly to the residents.

Self-driving 18-wheelers have longtime truckers worried about their livelihood and others concerned that the technology needs more testing to make sure the public is safe.

McDonald's concept restaurant CosMc's has taken its drink-focused menu to Dallas for its second-ever location.

With the country on the cusp of greeting the return of spring, a warm-weather treat is once again available for free for a limited time only.

Kelli and Michael Regan were looking for a new dog. The breeder they found online asked them to pay with gift cards.

Target, looking for ways to add sales, is relaunching its Target Circle loyalty program including a new paid membership with unlimited free same-day delivery in as little as an hour for orders over $35.

The $872 million most likely excludes any amount UnitedHealth may have paid to hackers in ransom.

George Schappell and sister Lori, of Reading, Pa., were the world's oldest conjoined twins, according to the Guinness Book of World Records.

Most worrisome gaps involve cancer chemotherapy drugs, ER medications and and therapies for ADHD.

The prepackaged boxes of deli meat, cheese and crackers are not a healthy choice for kids, advocacy group says.

This marks only the second-ever case of bird flu in humans in the U.S.

The projects are expected to create at least 17,000 construction jobs and 4,500 manufacturing jobs.

After more than 40 years in business, 99 Cents Only Stores, a discount chain, announced on Thursday that it will close all 371 of its locations and cease operations.

"This is huge, HUGE! If we don't plan appropriately, A, we won't have workers. Or B, we'll have so many people on the streets that nobody can get to the events."

"This is going to be an event all the way through the weekend, even starting as early as Friday."

Federal officials say milk from dairy cows in Texas and Kansas has tested positive for bird flu.

Michael Lorenzen pitched five spotless innings in his Texas debut and three relievers completed a five-hit shutout as the Rangers topped the Detroit Tigers 1-0.

Caitlin Clark has been selected with the No. 1 pick in the WNBA draft by the Indiana Fever.

"Scottie's a very humble guy. Wonderful personality and a great representative for Highland Park and for Dallas."

World no. 1 Scottie Scheffler finished 11 under to win his second Masters Tournament in three years Sunday.

Here's how much the winner took home last year, and what other golfers made.

In the 1,000th episode, titled "A Thousand Yards," NCIS comes under attack by a mysterious enemy from the past.

A Billy Joel special on CBS and Paramount+ will air again after it was cut off in the middle of the singer's performance of "Piano Man."

A judge in Texas is to hear arguments in rap star Travis Scott's request to be dismissed from a lawsuit over the deadly 2021 Astroworld festival in Houston.

Eleanor documented much of the chaos on "Apocalypse Now" in what would become one of the most famous making-of films about moviemaking, 1991's "Hearts of Darkness: A Filmmaker's Apocalypse."

CBS announces "The Talk" is ending its award-winning run in December with a shortened season 15.

The City of North Richland Hills says drone flights could begin as early as Monday of next week. The drone deliveries are a partnership between Walmart and a company called Wing and will be offered in North Richland Hills and surrounding cities. Other cities in North Texas, like Rrisco and Little Elm, already offer similar drone delivery.

Temperature will approach 90 degrees Thursday before a cold front passes through. Strong to severe storms are possible.

Almost every day, someone is killed in the U.S. during a high-speed police chase.

It took just one day for Texas to appeal a federal judge's ruling concerning the state's foster care system. A federal judge is fining the state $100,000 a day for contempt and neglecting to adequately investigate allegations of abuse against some who are in the system. As of February, there are about 10,000 children in the system statewide. An estimated 789 boys and girls are in foster care in Dallas County, and 784 in Tarrant County as of April 16, 2024.

Following Frederick Haynes' resignation, Rev. Jesse Jackson said his son, Yusef Jackson will continue to serve as Chief Operating Officer.

Dallas artist Roberto Marquez traveled to the Rafah Crossing in Egypt, the U.S. capital and will attend this weekend's statewide protest in Austin.

On Friday, hundreds of thousands of fans gathered outside and all around Globe Life Field in Arlington to celebrate the Texas Rangers historical World Series win!

Babies in the neonatal intensive care unit at several Texas Health hospitals were dressed in creative costumes for Halloween.

Is that the smell of cotton candy, beignets and brisket wafting over Fair Park? It sure is, and we are here for it!

No one puts these dolls back in their boxes. Babies in the neonatal intensive care unit at Texas Health Harris Methodist Hospital Southwest Fort Worth are pretty in pink!