CBS News Live
CBS News Texas: Local News, Weather & More
Watch CBS News





Texas state law says a child under the age of 10 doesn't have criminal culpability, law enforcement said.

A deep-dive into the Texas-sized Dallas bond election.
According to the Southlake Police Department, six cows in possible contact with the deceased animal have been quarantined.

The man remains in custody after being charged with 13 counts of theft of a firearm/enhanced.

A Billy Joel special on CBS and Paramount+ will air again after it was cut off in the middle of the singer's performance of "Piano Man."

The inmate had been in custody since Dec. 4, 2023, after being arrested by Fort Worth police for unlawful possession of a firearm by a felon and deadly conduct.

New safety policies like more lighting and a heavier police presence have been implemented in the West 7th cultural district.

A family says that on city time, in a city vehicle, and as a city camera recorded, two sanitation workers allegedly stole a pricey dog.

Girls Inc. hosted an event to teach young women about car maintenance basics.

April is Second Chance Month, recognizing the thousands of Americans being released from prisons and jails each day.

Dallas megachurch pastor Dr. Frederick Haynes III will step down as president and CEO of Rainbow PUSH Coalition.

Dallas' mayor is also calling for city council members to agree not to include a golden parachute clause in the next city manager's contract.
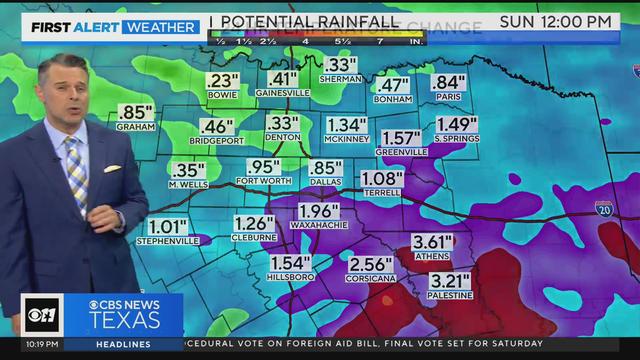
Be prepared for rain Saturday and then cold mornings Sunday and Monday.

For the first time in 30 years, Texas has a new lake: Bois D'Arc Lake in Fannin County.

A Dallas family says sanitation workers picked up more than trash. They say they "dognapped" their pet.

Netflix has gained more than 9 million subscribers since it cracked down on users sharing passwords.

College students, using a 3D printer, hope to give Polly the Duck a brand-new prosthetic bill.
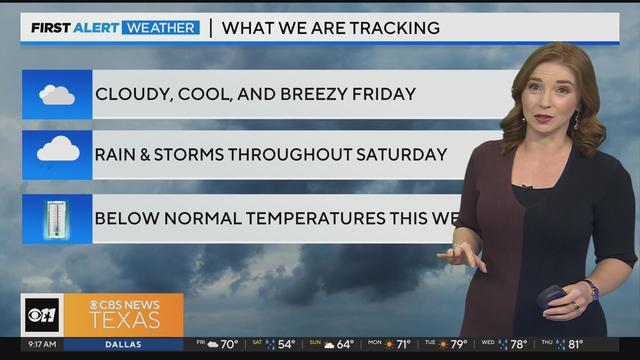
Friday will be cloudy and cool, and breezy but still dry for the most part. A lot of North Texas won't see rain until overnight hours into Saturday.
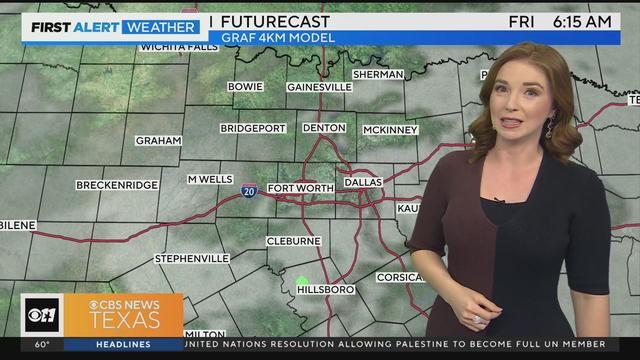
We are expecting mostly cloudy skies today, but dry conditions until the overnight hours.

We are expecting mostly cloudy skies today, but dry conditions until the overnight hours.

Texas police departments have the discretion to determine the frequency and extent of additional driving training for their officers. While some require driving training yearly or every other year, others do not.

Some departments opt to melt the firearms down, while others choose to crush them. However, there are instances where firearms, or at least parts of them, escape destruction altogether.

Several police departments told the CBS News Texas I-Team they were unaware of this practice, even though it was stated in the contracts they signed with the company, Gulf Coast GunBusters.

It's a complicated process that not everyone qualifies for.

Tuesday, federal prosecutors called their first witnesses against Dr. Raynaldo Ortiz.

The Dallas Cowboys have signed free agent running back Royce Freeman.

An attorney for the accuser says they plan to refile the lawsuit in Collin County.

Michael Lorenzen pitched five spotless innings in his Texas debut and three relievers completed a five-hit shutout as the Rangers topped the Detroit Tigers 1-0.

Caitlin Clark has been selected with the No. 1 pick in the WNBA draft by the Indiana Fever.

"Scottie's a very humble guy. Wonderful personality and a great representative for Highland Park and for Dallas."

Texas state law says a child under the age of 10 doesn't have criminal culpability, law enforcement said.

A deep-dive into the Texas-sized Dallas bond election.

According to the Southlake Police Department, six cows in possible contact with the deceased animal have been quarantined.

The man remains in custody after being charged with 13 counts of theft of a firearm/enhanced.

A Billy Joel special on CBS and Paramount+ will air again after it was cut off in the middle of the singer's performance of "Piano Man."

Texas police departments have the discretion to determine the frequency and extent of additional driving training for their officers. While some require driving training yearly or every other year, others do not.

Some departments opt to melt the firearms down, while others choose to crush them. However, there are instances where firearms, or at least parts of them, escape destruction altogether.

Several police departments told the CBS News Texas I-Team they were unaware of this practice, even though it was stated in the contracts they signed with the company, Gulf Coast GunBusters.

It's a complicated process that not everyone qualifies for.

Tuesday, federal prosecutors called their first witnesses against Dr. Raynaldo Ortiz.

Former Dallas council member, Jennifer Staubach Gates says, ""If you want a great police department, it starts with the training, and this is going to help us retain and recruit, which we desperately need."

Tamara Hutcherson with Stop Cop City Dallas says, "We believe that will result in more over-spending and repression in our communities and we think that money could be utilized elsewhere."
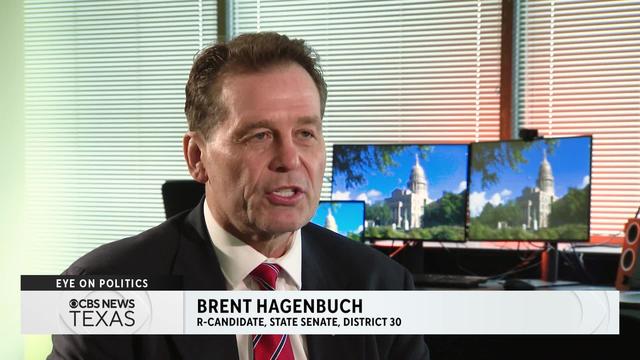
Brent Hagenbuch, a former Denton County GOP Chair, emerged to compete in the May 28 runoff for the 30th District State Senate seat.

A deep-dive into the Texas-sized Dallas bond election.

Dallas Council Member, Paula Blackmon discusses why she supports the $1.25 Billion 2024 Dallas Bond. Blackmon said, "We need to invest in our city, plain and simple."

Self-driving 18-wheelers have longtime truckers worried about their livelihood and others concerned that the technology needs more testing to make sure the public is safe.

McDonald's concept restaurant CosMc's has taken its drink-focused menu to Dallas for its second-ever location.

With the country on the cusp of greeting the return of spring, a warm-weather treat is once again available for free for a limited time only.

Kelli and Michael Regan were looking for a new dog. The breeder they found online asked them to pay with gift cards.

Target, looking for ways to add sales, is relaunching its Target Circle loyalty program including a new paid membership with unlimited free same-day delivery in as little as an hour for orders over $35.

The CDC estimates the U.S. could reach 300 measles cases in 2024 — more than the recent peak two years ago.

The $872 million most likely excludes any amount UnitedHealth may have paid to hackers in ransom.

George Schappell and sister Lori, of Reading, Pa., were the world's oldest conjoined twins, according to the Guinness Book of World Records.

Most worrisome gaps involve cancer chemotherapy drugs, ER medications and and therapies for ADHD.

The prepackaged boxes of deli meat, cheese and crackers are not a healthy choice for kids, advocacy group says.

The projects are expected to create at least 17,000 construction jobs and 4,500 manufacturing jobs.

After more than 40 years in business, 99 Cents Only Stores, a discount chain, announced on Thursday that it will close all 371 of its locations and cease operations.

"This is huge, HUGE! If we don't plan appropriately, A, we won't have workers. Or B, we'll have so many people on the streets that nobody can get to the events."

"This is going to be an event all the way through the weekend, even starting as early as Friday."

Federal officials say milk from dairy cows in Texas and Kansas has tested positive for bird flu.

The Dallas Cowboys have signed free agent running back Royce Freeman.

An attorney for the accuser says they plan to refile the lawsuit in Collin County.

Michael Lorenzen pitched five spotless innings in his Texas debut and three relievers completed a five-hit shutout as the Rangers topped the Detroit Tigers 1-0.

Caitlin Clark has been selected with the No. 1 pick in the WNBA draft by the Indiana Fever.

"Scottie's a very humble guy. Wonderful personality and a great representative for Highland Park and for Dallas."

Taylor Swift broke her own records, Spotify said, and now owns the record for the top three most-streamed albums in a single day.

The singer was found deceased at her home, a representative said.

Anticipation was growing at a fever pitch before Taylor Swift's latest album, "The Tortured Poets Department," dropped at midnight EDT. But it turned out it's actually a double album.

The singers first dated in 2003 and delighted fans when they rekindled their relationship in 2023.

Two country stars from Texas have partnered with a Nashville label to elevate Texas musicians.

Be prepared for rain Saturday and then cold mornings Sunday and Monday.

For the first time in 30 years, Texas has a new lake: Bois D'Arc Lake in Fannin County.

A Dallas family says sanitation workers picked up more than trash. They say they "dognapped" their pet.

Netflix has gained more than 9 million subscribers since it cracked down on users sharing passwords.

College students, using a 3D printer, hope to give Polly the Duck a brand-new prosthetic bill.

Dallas artist Roberto Marquez traveled to the Rafah Crossing in Egypt, the U.S. capital and will attend this weekend's statewide protest in Austin.

On Friday, hundreds of thousands of fans gathered outside and all around Globe Life Field in Arlington to celebrate the Texas Rangers historical World Series win!

Babies in the neonatal intensive care unit at several Texas Health hospitals were dressed in creative costumes for Halloween.

Is that the smell of cotton candy, beignets and brisket wafting over Fair Park? It sure is, and we are here for it!

No one puts these dolls back in their boxes. Babies in the neonatal intensive care unit at Texas Health Harris Methodist Hospital Southwest Fort Worth are pretty in pink!