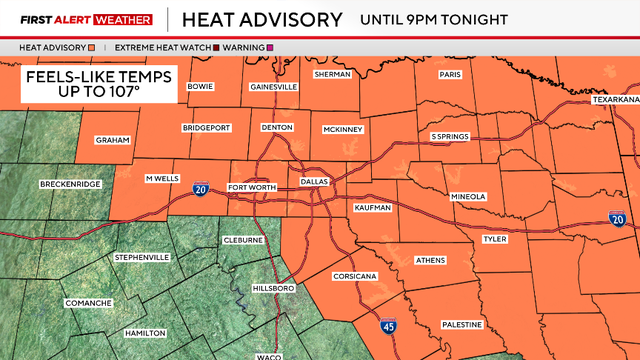
Heat advisory, First Alert Weather Day continues in North Texas
The heat advisory issued by the National Weather Service is in effect until 9 p.m. for most of North Texas.
Watch CBS News

The heat advisory issued by the National Weather Service is in effect until 9 p.m. for most of North Texas.

After the floods that impacted Central Texas on July 4, 39 local and national sports organizations came together to launch the Texas Sports Healing Fund.

As Central Texas recovers from deadly floods, CBS Texas is hosting the "Loud for Love" concert to support victims.

The break-in happened just after 3:10 a.m. in the 1400 block of E. Maddox Ave.

With little say in the ongoing debate over redrawing congressional districts in their home state, Texas Democratic state legislators are looking beyond their borders for help.

Five people were injured in the crash.

The collie was diagnosed with glaucoma after experiencing discomfort and cloudiness in her right eye.

The man claimed he was ridiculed after his bare behind was splashed over the internet for all to see.

The school district notified parents about the subpoena via letter on July 24.





Several organizations are accepting donations for those impacted by the floods, as well as first responders and volunteers.

After the floods that impacted Central Texas on July 4, 39 local and national sports organizations came together to launch the Texas Sports Healing Fund.
The Dallas entrepreneur is teaming up with local groups to support victims and first responders after deadly Central Texas flooding.

Top state officials are testifying that communication needs to improve between the state and local officials, between first responders, and with the large number of volunteers on the ground.

Seventeen-year-old Charlotte March wrote a song about her love for summer camp, then turned it into a fundraiser for families devastated by the Hill Country floods.

For the first time, we're seeing video that shows a Grand Prairie city councilman allegedly assaulting a real estate broker, which led to his arrest.

Gov. Greg Abbott explicitly asked lawmakers to regulate, not ban, hemp products.

While a North Texas man nearly cut in half by a boat during a family trip to the Bahamas remains hospitalized in Miami, his wife is urging Bahamian authorities to arrest and charge those responsible.
Another heat advisory and First Alert Weather Day are in effect on Friday due to dangerous heat lingering around North Texas.

Maxwell is currently serving a 20-year sentence at a low-security federal correctional institute in Tallahassee. She was convicted in 2021 for her role in helping Epstein recruit, groom and abuse underage girls.

Canine Companions pup, Lonestar, practiced his skills with a new person in the latest pupdate as we follow his journey to becoming a service dog. In a few short weeks, he will be matched with his permanent handler.
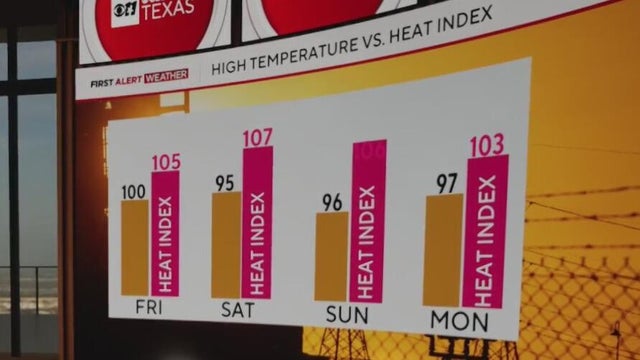
On Friday, the heat index will bring "feels-like" temperatures to 105 degrees.

An investigation is underway on Grapevine Mills Parkway after a crash involving multiple vehicles.

Another heat advisory and First Alert Weather Day are in effect on Friday due to dangerous heat lingering around North Texas.

The heat advisory issued by the National Weather Service is in effect until 9 p.m. for most of North Texas.

On Friday, the heat index will bring "feels-like" temperatures to 105 degrees.

Thursday marked the hottest day of the year so far for the DFW metroplex, as our high temperature topped out at 98° at DFW Airport.

For migrant children, who range in age from 3 to 17 years old and arrive in the U.S. without a guardian, applying for asylum can be a confusing obstacle.

SB 36 will create a division of Homeland Security within Texas's Department of Public Safety.

Two weeks ago, the Trump administration began carrying out its latest tactic aimed at fast-tracking deportations.

The detentions come on the heels of similar arrests earlier this week in immigration courts across the country.

Multiple sources have confirmed that at least a handful of people were arrested on the spot after their cases were dismissed in Dallas.

While illegal migrant crossings have dropped, immigration courts now have a historically high volume of cases

With the expansion of the 287(g) program, local and state officers will be able to enforce some immigration duties.

While the Trump administration says they're only targeting criminals for deportation, those words have done little to comfort some immigrants

A few weeks into his second term, President Donald Trump has issued dozens of executive orders.

After the floods that impacted Central Texas on July 4, 39 local and national sports organizations came together to launch the Texas Sports Healing Fund.

At the start of training camp, Cowboys head coach Brian Schottenheimer said his expectations in Oxnard are for the team to have "physical a-- practices."

Jonah Heim hit a looping fly ball that dropped between two fielders in shallow left field, scoring Wyatt Langford from first base, and the Texas Rangers beat the Athletics 2-1 on Wednesday night for a sweep.

Around this time last year, Cowboys star wide receiver CeeDee Lamb was holding out for a contract extension and was not in attendance for the 2024 training camp.

Arike Ogunbowale scored 20 points including four 3-pointers, Paige Bueckers had 14 points for her 19th straight double-digit game to begin her career, and the Dallas Wings beat the short-handed Seattle Storm 87-63.
Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

The heat advisory issued by the National Weather Service is in effect until 9 p.m. for most of North Texas.

After the floods that impacted Central Texas on July 4, 39 local and national sports organizations came together to launch the Texas Sports Healing Fund.

As Central Texas recovers from deadly floods, CBS Texas is hosting the "Loud for Love" concert to support victims.

The break-in happened just after 3:10 a.m. in the 1400 block of E. Maddox Ave.

With little say in the ongoing debate over redrawing congressional districts in their home state, Texas Democratic state legislators are looking beyond their borders for help.

The nonprofit employs men and women who have had past run-ins with the law to work as mentors for at-risk teens and young men.

Sidhartha "Sammy" Mukherjee and his wife Sunita became known for their Bollywood-style performances, became local celebrities, hosting parties and headlining music and cultural events.

On Thursday, the Texas House and Senate announced the creation of committees on disaster preparedness and flooding.

In what experts call "Flash Flood Alley," the terrain reacts quickly to rainfall steep slopes, rocky ground, and narrow riverbeds leave little time for warning.

For days before catastrophic floods left parts of Central Texas inundated, the National Weather Service was tracking the chance of rain.

With little say in the ongoing debate over redrawing congressional districts in their home state, Texas Democratic state legislators are looking beyond their borders for help.

The United States and Mexico have signed an agreement outlining specific steps to clean up the longstanding problem of Tijuana River sewage pouring across the border and polluting California beaches, officials say.

The effort to redraw the congressional boundaries could have major consequences on local, state and national politics.

The Texas attorney general is running in the GOP primary for U.S. Senate, and in the middle of a divorce from his wife, state Sen. Angela Paxton.

Deputy Attorney General Todd Blanche met with Ghislaine Maxwell in Florida about the case involving convicted sex offender Jeffrey Epstein.
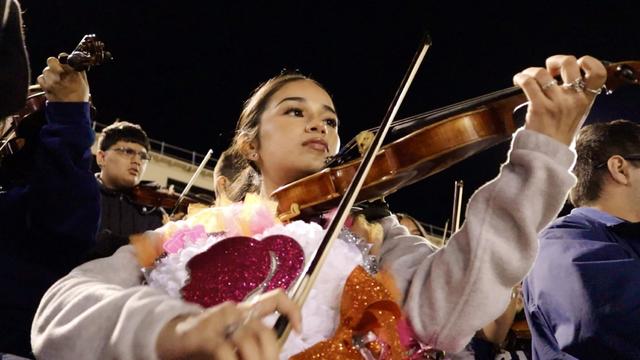
At Fort Worth ISD's North Side High School, the sounds of an American sport on the field meet mariachi music in the stands.

With new foods, such as the "Drowning Taquitos" and the "Beso de Angel," Tony's Taco Shop owners say they don't take their success for granted.

Latinas in Tech DFW started back up last year after the pandemic. They have lots of opportunities for Latinas to network, connect, and learn new skills.

Anchor Ken Molestina shows us how he makes his Cuban coffee for the CBS News Texas newsroom.

Del Olmo, who has played golf his entire life, recalls how rare the sport was for people like him growing up in Mexico City.

Less than two days after Delta Air Lines offered $30,000 to each passenger on board the flight that crashed and flipped in Toronto on Monday afternoon, the company is facing its first two lawsuits in the incident — and they likely won't be the last.

Texas Agricultural Commissioner Sid Miller is calling for a statewide ban on non-water additives, such as fluoride, in the public water system.

Last year, over 16 million vehicles drove on North Texas toll roads without paying, accumulating more than $69 million in unpaid tolls.

Activists are calling for a nationwide boycott of Target stores following the company's decision to roll back its diversity, equity and inclusion initiatives.

Discount store chain Target says it's joining rival Walmart and a number of other prominent American brands in scaling back corporate diversity, equity and inclusion initiatives.

Many Southeast, Southern and West Coast states are likely seeing an increase in COVID cases.

It's therapy that doesn't exactly look like therapy: outdoors, alongside a horse. Across North Texas, the demand for it is soaring.

Scientists conducting medical research are facing an existential crisis: Layoffs and budget cuts pushed by President Trump that, they say, jeopardize finding a cure for cancer.

A Dallas mail carrier died Saturday after collapsing on his mail route on the first day of summer.

Pest control company Terminix used call data to determine which cities had the most bed bug-infested areas in the country.

A truck had smashed through the front of Bruno's Place, destroying much of the inside.

Kroger said it intentionally decreased prices, but some customers said the financial drop at the register went in a different direction.

Could the solution to school shootings be drone first responders? An Austin-based tech company thinks so and is testing them at a private high school in Aurora.

Located off the Guadalupe River in Historic Old Ingram Loop, the shop was once filled floor to with lighting, decor and trinkets.

They were already concerned about tariffs, but construction businesses in North Texas said they have new fears with the president's immigration crackdown.

After the floods that impacted Central Texas on July 4, 39 local and national sports organizations came together to launch the Texas Sports Healing Fund.

At the start of training camp, Cowboys head coach Brian Schottenheimer said his expectations in Oxnard are for the team to have "physical a-- practices."

Jonah Heim hit a looping fly ball that dropped between two fielders in shallow left field, scoring Wyatt Langford from first base, and the Texas Rangers beat the Athletics 2-1 on Wednesday night for a sweep.

Around this time last year, Cowboys star wide receiver CeeDee Lamb was holding out for a contract extension and was not in attendance for the 2024 training camp.

Arike Ogunbowale scored 20 points including four 3-pointers, Paige Bueckers had 14 points for her 19th straight double-digit game to begin her career, and the Dallas Wings beat the short-handed Seattle Storm 87-63.

Paramount Global and Skydance Media agreed to merge last year.

Wrestling icon Hulk Hogan has died, police and World Wrestling Entertainment said Thursday.

Ozzy Osbourne rose to fame in the heavy metal group Black Sabbath.

The Alamo has acquired Pee-wee Herman's iconic bike from the 1985 film "Pee-wee's Big Adventure."

Actor and director Malcolm-Jamal Warner, best known for his role on "The Cosby Show," has died.

A suspect was taken into custody after an attack on Pearl Street Mall in Boulder on June 1 in which there were 15 people and a dog who were victims. The suspect threw Molotov cocktails that burned some of the victims, who were part of a march for Israeli hostages.

The Neonatal Intensive Care Unit babies at Texas Health locations across North Texas celebrated Valentine's Day.

As Anthony Davis prepared for his debut game at the AAC, Dallas Mavericks fans took to the arena to protest the controversial trade.

A look back at the esteemed personalities who've left us this year, who'd touched us with their innovation, creativity and humanity.

CBS News Texas viewers got out and enjoyed the snow day on Thursday and send us all of their best photos. Take a look.