CBS News Live
CBS News Texas: Local News, Weather & More
Watch CBS News





Think your alma mater has the prettiest campus in Texas? You might be right.

The vinyl sales alone were monumental, Billboard said, with "the largest sales week for an album on vinyl in the modern era."

The Dallas Cowboys and running back Ezekiel Elliott have agreed to terms pending a physical, NFL Network Insiders Ian Rapoport and Tom Pelissero first reported Monday morning.

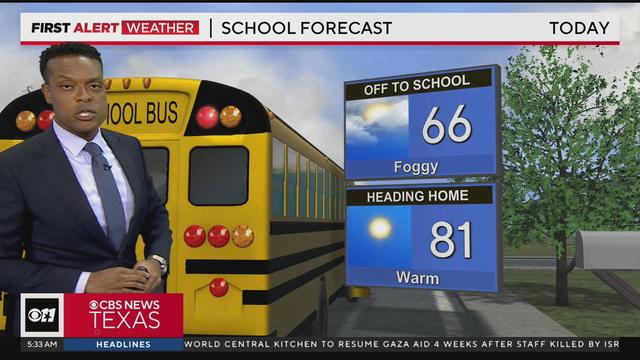
Highs will be in the mid-80s. Tonight, we'll see mostly clear skies with low temperatures in the lower 60s.

With multiple rounds of storms, some residents have had their homes flooded multiple times this weekend.

Game 4 is Monday night!

Monday and Tuesday look warm and storm-free. It will be breezy on Tuesday and a little more humid.

Sadly, a truck driver was killed when his 18-wheeler overturned.

The Los Angeles Clippers beat the Dallas Mavericks 116-111 on Sunday, evening the first-round series at 2-2.

Both Senator Ted Cruz, R-Texas and U.S. Rep. Colin Allred, D-Dallas are running for U.S. Senate this fall, which is the marquee race in Texas.

These are the airports Southwest is pulling out of completely as it looks to save costs.

Despite her experience, the Forest Hill woman tells CBS News Texas she's still Beverly's No. 1 fan and hopes her story is a warning to others.

Rain goes away for a little bit before making a return to North Texas.
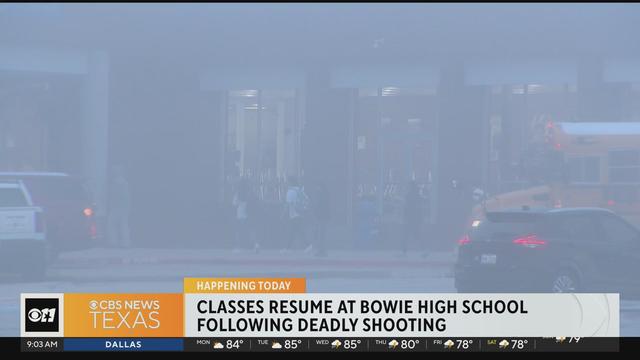
There was a heavy police presence when students returned to class Monday morning, five days after a student was fatally shot.
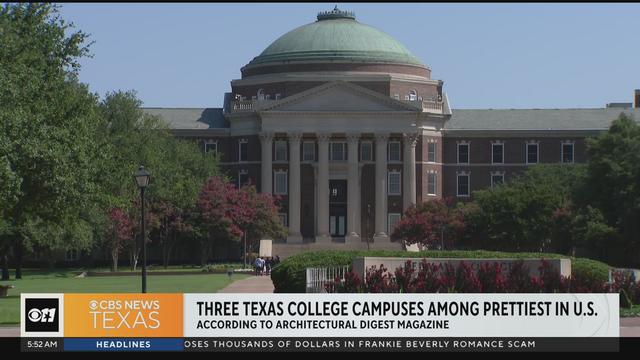
Architectural Digest Magazine says three Texas colleges are among the prettiest in the country. Did your alma mater make the list?!

Part of Dallas' longstanding history is going to be made available for purchase Monday. Antiques, furnishings and more can be yours starting at 10 a.m.

Four people died as a result of the tornadoes this weekend. Oklahoma's governor said the damage is some of the worst he's ever seen.

Rain goes away for a little bit before making a return to North Texas.
Make sure to slow down and use your low beam headlights!

Highs will be in the mid-80s. Tonight, we'll see mostly clear skies with low temperatures in the lower 60s.

A scammer a North Texas woman met on Instagram claimed to be a German cardiologist, and for months, the two messaged back and forth, building what she thought was a true relationship.

Texas police departments have the discretion to determine the frequency and extent of additional driving training for their officers. While some require driving training yearly or every other year, others do not.

Some departments opt to melt the firearms down, while others choose to crush them. However, there are instances where firearms, or at least parts of them, escape destruction altogether.

Several police departments told the CBS News Texas I-Team they were unaware of this practice, even though it was stated in the contracts they signed with the company, Gulf Coast GunBusters.

It's a complicated process that not everyone qualifies for.

The Dallas Cowboys and running back Ezekiel Elliott have agreed to terms pending a physical, NFL Network Insiders Ian Rapoport and Tom Pelissero first reported Monday morning.

Game 4 is Monday night!

The Los Angeles Clippers beat the Dallas Mavericks 116-111 on Sunday, evening the first-round series at 2-2.
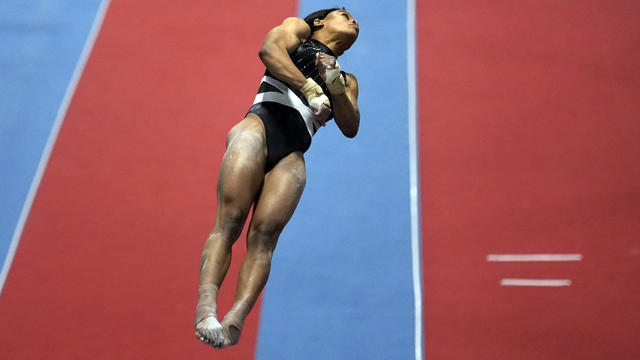
Gabby Douglas qualified in multiple events for the U.S. Championships in Fort Worth, Texas, next month.

Here's a look at who the 'Boys have drafted so far.

Think your alma mater has the prettiest campus in Texas? You might be right.

The vinyl sales alone were monumental, Billboard said, with "the largest sales week for an album on vinyl in the modern era."

The Dallas Cowboys and running back Ezekiel Elliott have agreed to terms pending a physical, NFL Network Insiders Ian Rapoport and Tom Pelissero first reported Monday morning.

Highs will be in the mid-80s. Tonight, we'll see mostly clear skies with low temperatures in the lower 60s.

With multiple rounds of storms, some residents have had their homes flooded multiple times this weekend.

A scammer a North Texas woman met on Instagram claimed to be a German cardiologist, and for months, the two messaged back and forth, building what she thought was a true relationship.

They found him guilty – now four jurors are explaining how they were convinced to convict Dr. Raynaldo Ortiz.

Texas police departments have the discretion to determine the frequency and extent of additional driving training for their officers. While some require driving training yearly or every other year, others do not.

Some departments opt to melt the firearms down, while others choose to crush them. However, there are instances where firearms, or at least parts of them, escape destruction altogether.

Several police departments told the CBS News Texas I-Team they were unaware of this practice, even though it was stated in the contracts they signed with the company, Gulf Coast GunBusters.
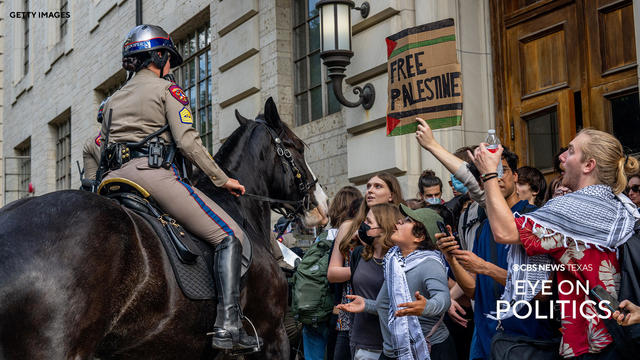
Every week, Eye on Politics breaks down some of the biggest political stories grabbing headlines in North Texas and beyond.
Dozens of people were arrested at the University of Texas at Austin during protests over the Israel-Hamas war and state lawmakers are divided on the university's response.

Senator Ted Cruz explains why he voted against a bill to give funds to Israel and discusses his opinion on protests happening on college campuses across the U.S.
Democratic Congressman and Democratic nominee for U.S. Senate, Colin Allred, discusses his plans moving forward in his campaign for Texas Senate against incumbent Ted Cruz.

Both Senator Ted Cruz, R-Texas and U.S. Rep. Colin Allred, D-Dallas are running for U.S. Senate this fall, which is the marquee race in Texas.

Self-driving 18-wheelers have longtime truckers worried about their livelihood and others concerned that the technology needs more testing to make sure the public is safe.

McDonald's concept restaurant CosMc's has taken its drink-focused menu to Dallas for its second-ever location.

With the country on the cusp of greeting the return of spring, a warm-weather treat is once again available for free for a limited time only.

Kelli and Michael Regan were looking for a new dog. The breeder they found online asked them to pay with gift cards.

Target, looking for ways to add sales, is relaunching its Target Circle loyalty program including a new paid membership with unlimited free same-day delivery in as little as an hour for orders over $35.

The White House had been due to decide on the menthol cigarette rule in March.
First known HIV cases from a nonsterile injection for cosmetic reasons highlights the risk of unlicensed providers.

The CDC estimates the U.S. could reach 300 measles cases in 2024 — more than the recent peak two years ago.

Organic option is best when buying certain produce, especially blueberries, nonprofit group says in analysis of chemical residues.

The $872 million most likely excludes any amount UnitedHealth may have paid to hackers in ransom.

Within hours of the vote, the U.S. Chamber of Commerce announced it would sue to block the ban. Dallas employment attorney Rogge Dunn predicts employees will ultimately win this battle.

The closure affects both Dom's locations in Chicago, and all 33 Foxtrot stores in Chicago, Texas, and the Washington D.C. area.

Texas law SB 14 prohibits drug and surgical "gender transition" interventions for minors.

The projects are expected to create at least 17,000 construction jobs and 4,500 manufacturing jobs.

After more than 40 years in business, 99 Cents Only Stores, a discount chain, announced on Thursday that it will close all 371 of its locations and cease operations.

The Dallas Cowboys and running back Ezekiel Elliott have agreed to terms pending a physical, NFL Network Insiders Ian Rapoport and Tom Pelissero first reported Monday morning.

Game 4 is Monday night!

The Los Angeles Clippers beat the Dallas Mavericks 116-111 on Sunday, evening the first-round series at 2-2.
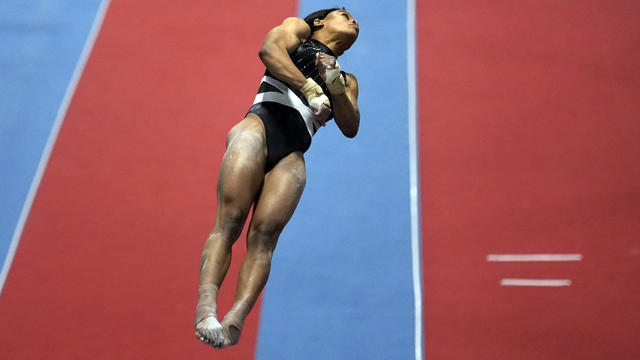
Gabby Douglas qualified in multiple events for the U.S. Championships in Fort Worth, Texas, next month.

Here's a look at who the 'Boys have drafted so far.

The vinyl sales alone were monumental, Billboard said, with "the largest sales week for an album on vinyl in the modern era."

Harvey Weinstein's 2020 conviction on felony sex crime charges has been overturned by the State of New York Court of Appeals.

Taylor Swift fans have found a way to feel "a little bit closer to" their hero at a London watering hole, and The Black Dog pub is lapping it up.

Mary J. Blige, Cher, Foreigner, A Tribe Called Quest, Kool & The Gang, Ozzy Osbourne, Dave Matthews Band and Peter Frampton have been named to the Rock & Roll Hall of Fame.

Taylor Swift broke her own records, Spotify said, and now owns the record for the top three most-streamed albums in a single day.

Rain goes away for a little bit before making a return to North Texas.

There was a heavy police presence when students returned to class Monday morning, five days after a student was fatally shot.

Architectural Digest Magazine says three Texas colleges are among the prettiest in the country. Did your alma mater make the list?!

Part of Dallas' longstanding history is going to be made available for purchase Monday. Antiques, furnishings and more can be yours starting at 10 a.m.

Four people died as a result of the tornadoes this weekend. Oklahoma's governor said the damage is some of the worst he's ever seen.

Dallas artist Roberto Marquez traveled to the Rafah Crossing in Egypt, the U.S. capital and will attend this weekend's statewide protest in Austin.

On Friday, hundreds of thousands of fans gathered outside and all around Globe Life Field in Arlington to celebrate the Texas Rangers historical World Series win!

Babies in the neonatal intensive care unit at several Texas Health hospitals were dressed in creative costumes for Halloween.

Is that the smell of cotton candy, beignets and brisket wafting over Fair Park? It sure is, and we are here for it!

No one puts these dolls back in their boxes. Babies in the neonatal intensive care unit at Texas Health Harris Methodist Hospital Southwest Fort Worth are pretty in pink!