
Texas House Democrats return to Austin as GOP redistricting push resumes
The Texas House is scheduled to try convening a quorum again at noon.
Watch CBS News

The Texas House is scheduled to try convening a quorum again at noon.

President Trump is set to meet with Ukrainian President Volodymyr Zelenskyy and European leaders on Monday as the U.S. pursues a peace agreement between Russia and Ukraine.

From the Metroplex to the east, a heat advisory is in place until 8 p.m. for feels-like temperatures as high as 107.

Former Attorney General William Barr is the first person scheduled to be deposed by Congress members as questions surrounding his role in the Epstein investigation persist.

Police say the alleged stolen items were linked to several recent vehicle burglaries.

Wilson leads red-hot Aces to seventh straight win as Bell and Gray shine in high-scoring victory over struggling Wings.

Authorities are working to identify those responsible for what Heath's mayor called a "hateful display" outside the school.

Marcus Semien homered and scored twice in his return to the lineup and Nathan Eovaldi worked seven innings and won his seventh straight decision as the Texas Rangers beat the Toronto Blue Jays 10-4 to avoid a three-game sweep.

The blaze began shortly after 2 p.m. and presented multiple challenges for firefighters.





Several organizations are accepting donations for those impacted by the floods, as well as first responders and volunteers.

Former campers, who are now adults, share their memories from Camp Mystic and remember those who died in the devastating Hill Country flood on July 4.

An act of nature has destroyed what was a kind gesture from a group of people in Parker County.

Texas officials have faced questions over their preparedness and the speed of their initial actions when flash flooding hit.

The public hearing will take place at Hill Country Youth Event Center at 9:30 a.m.

The 450,000-square-foot facility will support large-scale productions, job training, and boost the local economy.

FIFA needs more than 6,000 volunteers for the events in the Dallas area.
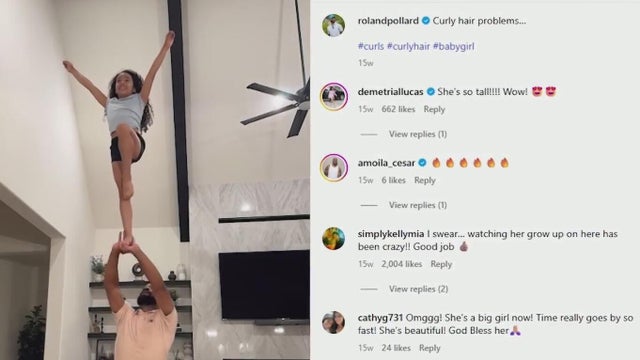
Cheer stunts, fatherly wisdom, and an unshakable bond — that's what you'll find on Roland Pollard's Instagram feed.
Hurricane Erin is not expected to hit the U.S. mainland, but its effects wil make swimming dangerous on the Atlantic coast.
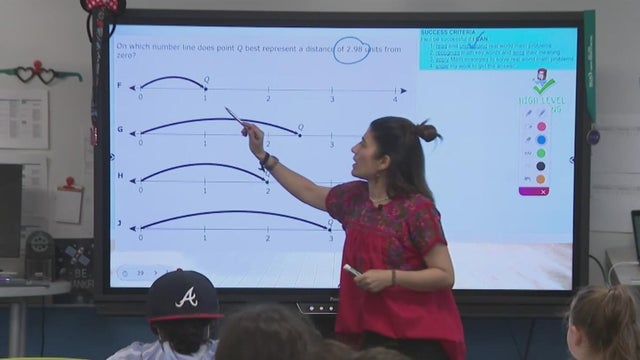
Two years ago, when a record 13% of teachers in Texas left their jobs, Gov. Greg Abbott commissioned a task force to look for solutions. Andrea Lucia and Lacey Beasley report on what they found.

Elliott's Voice installs color-coded, symbol-based communication boards across North Texas. Lauren Crawford reports.
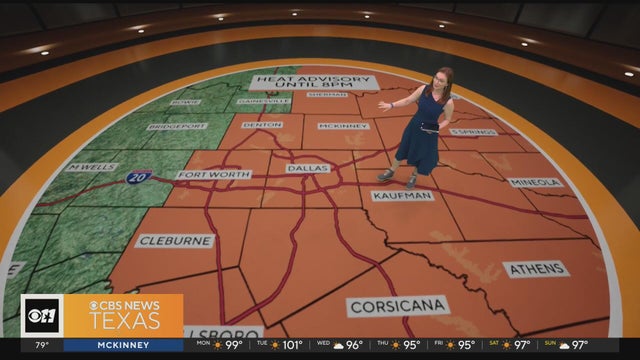
Parts of the Metroplex will have feels-like temperatures around 105 on Monday afternoon. First Alert meteorologist Brittany Rainey has the forecast.

The Texas House will attempt to reach quorum at noon Monday as the Republican majority pushes congressional redistricting. Trevor Sochocki reports.

From the Metroplex to the east, a heat advisory is in place until 8 p.m. for feels-like temperatures as high as 107.

Parts of the Metroplex will have feels-like temperatures around 105 on Monday afternoon. First Alert meteorologist Brittany Rainey has the forecast.

Hurricane Erin, which briefly reached Category 5 strength, battered parts of the Caribbean, including Puerto Rico and the U.S. Virgin Islands, with heavy rain and strong surf. Though it won’t make landfall in the U.S., the East Coast is facing dangerous rip currents and rough seas.
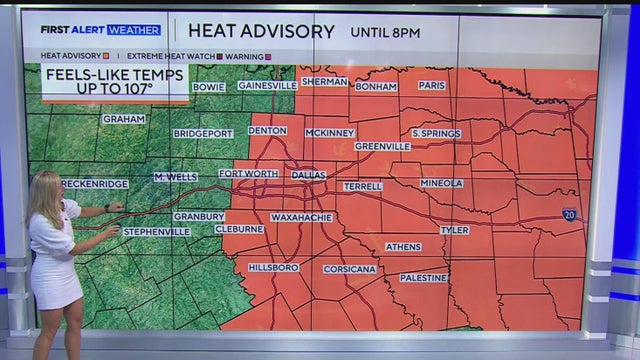
Another heat advisory is in the forecast for the week of Aug. 17 across North Texas. Some scattered storms and rain will move through the area mid-week.

For migrant children, who range in age from 3 to 17 years old and arrive in the U.S. without a guardian, applying for asylum can be a confusing obstacle.

SB 36 will create a division of Homeland Security within Texas's Department of Public Safety.

Two weeks ago, the Trump administration began carrying out its latest tactic aimed at fast-tracking deportations.

The detentions come on the heels of similar arrests earlier this week in immigration courts across the country.

Multiple sources have confirmed that at least a handful of people were arrested on the spot after their cases were dismissed in Dallas.

While illegal migrant crossings have dropped, immigration courts now have a historically high volume of cases

With the expansion of the 287(g) program, local and state officers will be able to enforce some immigration duties.

While the Trump administration says they're only targeting criminals for deportation, those words have done little to comfort some immigrants

A few weeks into his second term, President Donald Trump has issued dozens of executive orders.

The Rangers, held to four hits, led 2-1 after three innings on sacrifice flies by Adolis García and Marcus Semien.

Maddy Siegrist tied a career high with 22 points in her first start since June 6.

UFC CEO Dana White confirmed plans for a July 4, 2026 fight at the White House to mark America's 250th birthday.

Burger lined a 99.4 mph pitch from Andrew Saalfrank (0-1) off the left-field wall to end the game.

FIFA needs more than 6,000 volunteers for the events in the Dallas area.
Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

Eat See Play: Sponsored by Metroplex Cadillac Dealers

The Texas House is scheduled to try convening a quorum again at noon.

President Trump is set to meet with Ukrainian President Volodymyr Zelenskyy and European leaders on Monday as the U.S. pursues a peace agreement between Russia and Ukraine.

From the Metroplex to the east, a heat advisory is in place until 8 p.m. for feels-like temperatures as high as 107.

Former Attorney General William Barr is the first person scheduled to be deposed by Congress members as questions surrounding his role in the Epstein investigation persist.

Police say the alleged stolen items were linked to several recent vehicle burglaries.

Even though the project was first proposed 57 years ago, construction hasn't started on the Marvin Nichols Reservoir.

The VA in North Texas is using a new algorithm to calculate the hours of in-home care that veterans receive.

The nonprofit employs men and women who have had past run-ins with the law to work as mentors for at-risk teens and young men.

Sidhartha "Sammy" Mukherjee and his wife Sunita became known for their Bollywood-style performances, became local celebrities, hosting parties and headlining music and cultural events.

On Thursday, the Texas House and Senate announced the creation of committees on disaster preparedness and flooding.

The Texas House is scheduled to try convening a quorum again at noon.

President Trump is set to meet with Ukrainian President Volodymyr Zelenskyy and European leaders on Monday as the U.S. pursues a peace agreement between Russia and Ukraine.

Former Attorney General William Barr is the first person scheduled to be deposed by Congress members as questions surrounding his role in the Epstein investigation persist.

Texas Democratic lawmakers who fled the state to deny a quorum are making their way back, each on their own timeline, multiple sources familiar with the matter told CBS News.

Special U.S. envoy Steve Witkoff says Russian leader Vladimir Putin agreed to allow the U.S. and Europe to offer Ukraine a security guarantee resembling NATO's collective defense mandate.
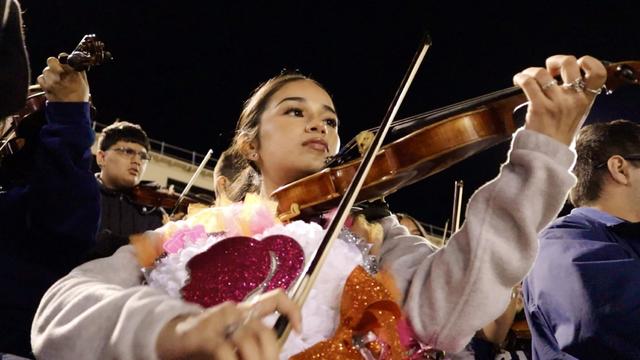
At Fort Worth ISD's North Side High School, the sounds of an American sport on the field meet mariachi music in the stands.

With new foods, such as the "Drowning Taquitos" and the "Beso de Angel," Tony's Taco Shop owners say they don't take their success for granted.

Latinas in Tech DFW started back up last year after the pandemic. They have lots of opportunities for Latinas to network, connect, and learn new skills.

Anchor Ken Molestina shows us how he makes his Cuban coffee for the CBS News Texas newsroom.

Del Olmo, who has played golf his entire life, recalls how rare the sport was for people like him growing up in Mexico City.

Less than two days after Delta Air Lines offered $30,000 to each passenger on board the flight that crashed and flipped in Toronto on Monday afternoon, the company is facing its first two lawsuits in the incident — and they likely won't be the last.

Texas Agricultural Commissioner Sid Miller is calling for a statewide ban on non-water additives, such as fluoride, in the public water system.

Last year, over 16 million vehicles drove on North Texas toll roads without paying, accumulating more than $69 million in unpaid tolls.

Activists are calling for a nationwide boycott of Target stores following the company's decision to roll back its diversity, equity and inclusion initiatives.

Discount store chain Target says it's joining rival Walmart and a number of other prominent American brands in scaling back corporate diversity, equity and inclusion initiatives.

Instagram rolled out a new Map feature that lets users see their friends' locations in real time. Here's why privacy experts are concerned.

For the first time in more than 30 years, a Fort Worth community long considered a "health care desert" now has nearby access to basic medical care.

Head Colorado Buffaloes football Coach Deion "Coach Prime" Sanders revealed his bladder cancer diagnosis for the first time on Monday morning.

Pepsi hops on another nutritional bandwagon as more consumers opt for prebiotic beverages.

Many Southeast, Southern and West Coast states are likely seeing an increase in COVID cases.

A truck had smashed through the front of Bruno's Place, destroying much of the inside.

Kroger said it intentionally decreased prices, but some customers said the financial drop at the register went in a different direction.

Could the solution to school shootings be drone first responders? An Austin-based tech company thinks so and is testing them at a private high school in Aurora.

Located off the Guadalupe River in Historic Old Ingram Loop, the shop was once filled floor to with lighting, decor and trinkets.

They were already concerned about tariffs, but construction businesses in North Texas said they have new fears with the president's immigration crackdown.

The Rangers, held to four hits, led 2-1 after three innings on sacrifice flies by Adolis García and Marcus Semien.

Maddy Siegrist tied a career high with 22 points in her first start since June 6.

UFC CEO Dana White confirmed plans for a July 4, 2026 fight at the White House to mark America's 250th birthday.

Burger lined a 99.4 mph pitch from Andrew Saalfrank (0-1) off the left-field wall to end the game.

FIFA needs more than 6,000 volunteers for the events in the Dallas area.

The Oscar-nominated actor famously played arch-villain General Zod in "Superman" and "Superman II."

Taylor Swift's upcoming album release and her love of all things orange isn't lost on her fans — or businesses clamoring for a piece of the action.

Taylor Swift shared several details about her new album, "The Life of a Showgirl," on a special episode of "New Heights," the podcast hosted by her boyfriend, Travis Kelce, and his brother, Jason Kelce.

The 450,000-square-foot facility will support large-scale productions, job training, and boost the local economy.

Swift joined "New Heights" alongside Travis Kelce, opening up about their relationship, buying back the master recordings of her first six albums, Easter eggs and her upcoming album.

A suspect was taken into custody after an attack on Pearl Street Mall in Boulder on June 1 in which there were 15 people and a dog who were victims. The suspect threw Molotov cocktails that burned some of the victims, who were part of a march for Israeli hostages.

The Neonatal Intensive Care Unit babies at Texas Health locations across North Texas celebrated Valentine's Day.

As Anthony Davis prepared for his debut game at the AAC, Dallas Mavericks fans took to the arena to protest the controversial trade.

A look back at the esteemed personalities who've left us this year, who'd touched us with their innovation, creativity and humanity.

CBS News Texas viewers got out and enjoyed the snow day on Thursday and send us all of their best photos. Take a look.