CBS News Live
CBS News Texas: Local News, Weather & More
Watch CBS News




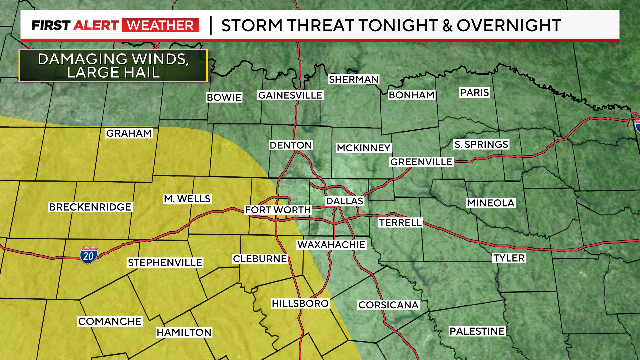
Flood Watch will remain in effect until 1 p.m. Sunday.

The U.S. Coast Guard said they had medevaced an hours-old baby from Cleveland, Texas, amid the floodwaters.

It was a Texas veterinarian who collected samples from dairy farms that confirmed the outbreak of H5N1 bird flu in cattle for the first time.

Voters could get transportation to polling locations a number of different ways.

An isolated shower and drizzle are possible from time to time with spotty afternoon storms.
In the harrowing live video from Freddy McKinney, he yells at the family to "Hurry, get inside!" as they run for their lives, begging him for help.
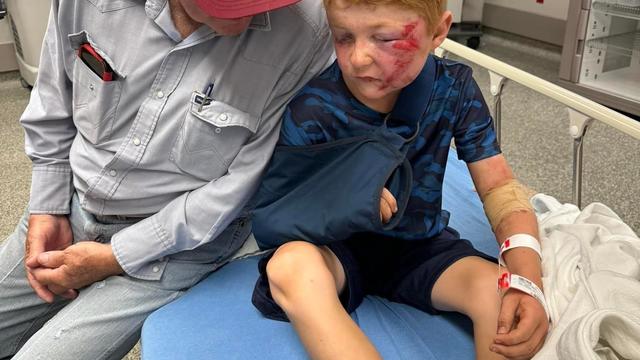
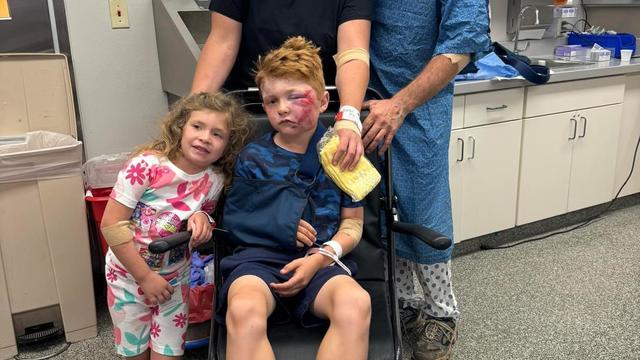
A storm chaser not only captured a massive tornado touching down in Hawley, Texas on Thursday but ended up rescuing a family of four whose home was destroyed by it.
Some business owners & managers say a lack of enforcement is driving away customers.

Nine-year-old Kelvin Ellis Jr. had just received the dollar for good grades, and it was the only money he had to his name.

Luka Doncic had 28 points and 13 assists, Kyrie Irving scored 28 of 30 points in a second-half surge and the Dallas Mavericks advanced to the second round of the playoffs with a 114-101 victory over the Los Angeles Clippers.

In the harrowing live video from Freddy McKinney, he yells at the family to "Hurry, get inside!" as they run for their lives, begging him for help.

The 97-year-old is one of 19 individuals who received the nation's highest civilian honor Friday.

The Keller High girls softball team didn't let the loss of numerous pieces of equipment get in the way of earning a playoff victory.
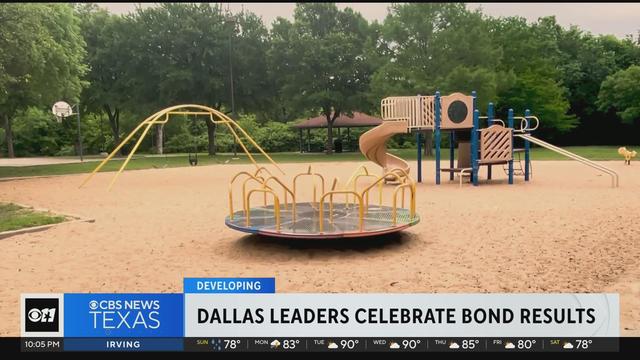
Officials in North Texas talk about results in Saturday's municipal elections.

CBS News Texas meteorologist Jeff Ray visited H.D. Staples Elementary School in Joshua as part of "Weather on Wheels."
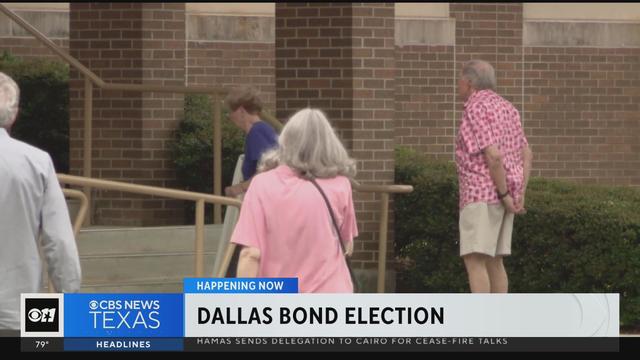
Voters headed to the polls Saturday for municipal elections.
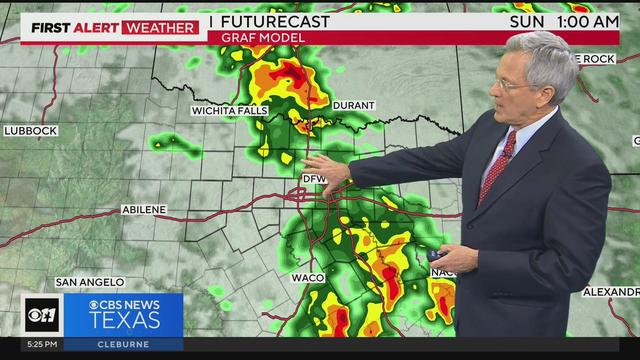
A Weather Alert has been issued for the overnight hours as rain and thunderstorms will move into North Texas from the west.
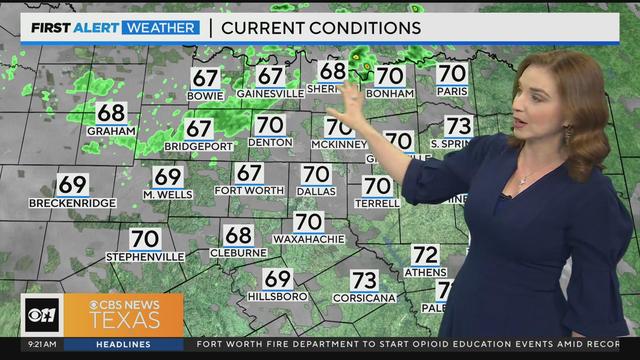
An isolated shower and drizzle are possible from time to time with spotty afternoon storms.

An isolated shower and drizzle are possible from time to time with spotty afternoon storms.
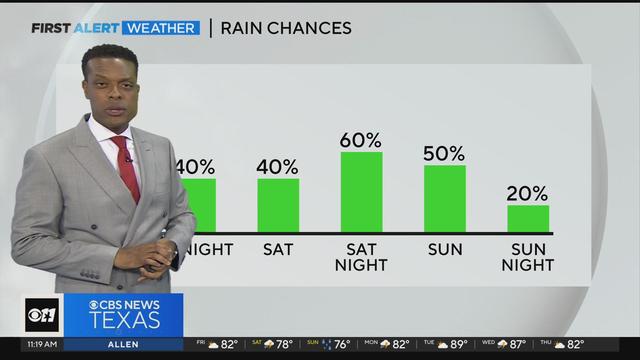
This weekend will not be a complete washout, but prepare for a chance of rain and storms. The best chance for heavier rain/storms will arrive Saturday night into Sunday morning. Some localized flooding is possible this weekend.
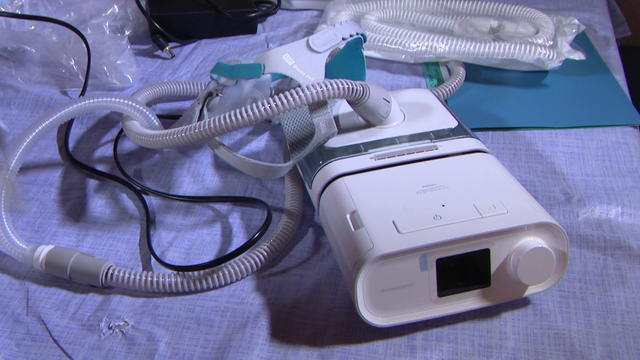
In a statement, Philips said it does not admit any wrongdoing but chose to settle "to end the uncertainty associated with litigation in the U.S."

A scammer a North Texas woman met on Instagram claimed to be a German cardiologist, and for months, the two messaged back and forth, building what she thought was a true relationship.

Texas police departments have the discretion to determine the frequency and extent of additional driving training for their officers. While some require driving training yearly or every other year, others do not.

Some departments opt to melt the firearms down, while others choose to crush them. However, there are instances where firearms, or at least parts of them, escape destruction altogether.

Several police departments told the CBS News Texas I-Team they were unaware of this practice, even though it was stated in the contracts they signed with the company, Gulf Coast GunBusters.

It was just the 10th Kentucky Derby decided by a nose, and the first since Grindstone wore the garland of red roses in 1996.

A Dallas police source said the sexual assault investigation is over due to insufficient evidence.

Luka Doncic scored 20 of his 35 points in the second half and added 10 assists and seven rebounds, propelling the Dallas Mavericks to a 123-93 victory over the Los Angeles Clippers in Game 5 and a 3-2 lead in the first-round series.

Jason Robertson scored a power-play goal to put Dallas ahead late in the second period after Tyler Seguin took a shot to the face and the Stars beat the Vegas Golden Knights 3-2 in Game 5.

The running back is officially a Dallas Cowboy again after a steak dinner that sealed the deal.

Flood Watch will remain in effect until 1 p.m. Sunday.

The U.S. Coast Guard said they had medevaced an hours-old baby from Cleveland, Texas, amid the floodwaters.

It was a Texas veterinarian who collected samples from dairy farms that confirmed the outbreak of H5N1 bird flu in cattle for the first time.

Voters could get transportation to polling locations a number of different ways.

An isolated shower and drizzle are possible from time to time with spotty afternoon storms.

In a statement, Philips said it does not admit any wrongdoing but chose to settle "to end the uncertainty associated with litigation in the U.S."

A scammer a North Texas woman met on Instagram claimed to be a German cardiologist, and for months, the two messaged back and forth, building what she thought was a true relationship.

They found him guilty – now four jurors are explaining how they were convinced to convict Dr. Raynaldo Ortiz.

Texas police departments have the discretion to determine the frequency and extent of additional driving training for their officers. While some require driving training yearly or every other year, others do not.

Some departments opt to melt the firearms down, while others choose to crush them. However, there are instances where firearms, or at least parts of them, escape destruction altogether.

State Rep. Ana-Maria Ramos of Dallas also tells political reporter Jack Fink she disagrees with the State of Texas' attempts to block new Title IX rules meant to protect LGBTQ+ students.
State Rep. Matt Shaheen of Plano also tells political reporter Jack Fink that he agrees with the State of Texas' attempts to block new Title IX rules meant to protect LGBTQ+ students.

Burns' challenger has received high-profile endorsements, including former President and presumed Republican Presidential nominee Donald Trump and Gov. Greg Abbott.

Ex-government employee Miguel Zapata is accused of sending fake FBI tips falsely accusing multiple coworkers of taking part in the Jan. 6, 2021, Capitol breach.
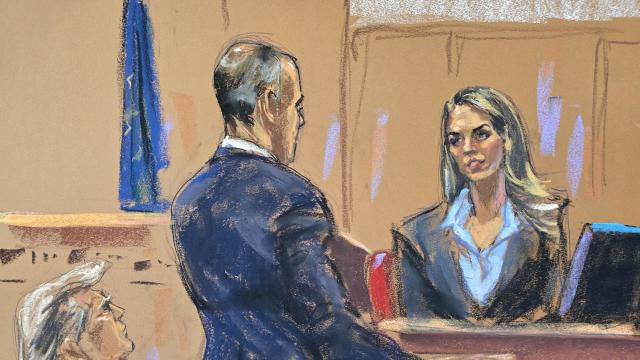
Hope Hicks, one of former President Donald Trump's closest aides for years, told jurors how she handled the fallout from "hush money" payments made to two women before the 2016 election.

Self-driving 18-wheelers have longtime truckers worried about their livelihood and others concerned that the technology needs more testing to make sure the public is safe.

McDonald's concept restaurant CosMc's has taken its drink-focused menu to Dallas for its second-ever location.

With the country on the cusp of greeting the return of spring, a warm-weather treat is once again available for free for a limited time only.

Kelli and Michael Regan were looking for a new dog. The breeder they found online asked them to pay with gift cards.

Target, looking for ways to add sales, is relaunching its Target Circle loyalty program including a new paid membership with unlimited free same-day delivery in as little as an hour for orders over $35.

The Texas dairy worker infected by H5N1 "did not disclose the name of their workplace," frustrating investigators.

A North Texas mother partnered with the Texas Health Resources Foundation to provide a potentially life-saving tool to at-risk pregnant and postpartum moms.

Plaintiffs have three months to vote on whether to approve a proposed legal settlement that would resolve nearly all talc lawsuits.

Cat deaths and neurological disease are "widely reported" around farms where the H5N1 bird flu virus was detected, health officials say.

The White House had been due to decide on the menthol cigarette rule in March.

Within hours of the vote, the U.S. Chamber of Commerce announced it would sue to block the ban. Dallas employment attorney Rogge Dunn predicts employees will ultimately win this battle.

The closure affects both Dom's locations in Chicago, and all 33 Foxtrot stores in Chicago, Texas, and the Washington D.C. area.

Texas law SB 14 prohibits drug and surgical "gender transition" interventions for minors.

The projects are expected to create at least 17,000 construction jobs and 4,500 manufacturing jobs.

After more than 40 years in business, 99 Cents Only Stores, a discount chain, announced on Thursday that it will close all 371 of its locations and cease operations.

It was just the 10th Kentucky Derby decided by a nose, and the first since Grindstone wore the garland of red roses in 1996.

A Dallas police source said the sexual assault investigation is over due to insufficient evidence.

Luka Doncic scored 20 of his 35 points in the second half and added 10 assists and seven rebounds, propelling the Dallas Mavericks to a 123-93 victory over the Los Angeles Clippers in Game 5 and a 3-2 lead in the first-round series.

Jason Robertson scored a power-play goal to put Dallas ahead late in the second period after Tyler Seguin took a shot to the face and the Stars beat the Vegas Golden Knights 3-2 in Game 5.

The running back is officially a Dallas Cowboy again after a steak dinner that sealed the deal.

The start of the first civil trial stemming from the deadly crowd surge at the 2021 Astroworld festival in Texas has been delayed.

"Happy 9th Birthday, Princess Charlotte!" the Prince and Princess of Wales said in a social media post with a new photo of their daughter taken by Kate.

Tickets are not officially on sale yet. You can sign up for presale tickets on the AT&T Stadium website.

Paramount said long-time CEO Bob Bakish will leave the company, which is in discussions to explore a sale or merger.

The vinyl sales alone were monumental, Billboard said, with "the largest sales week for an album on vinyl in the modern era."

The Keller High girls softball team didn't let the loss of numerous pieces of equipment get in the way of earning a playoff victory.

Officials in North Texas talk about results in Saturday's municipal elections.

CBS News Texas meteorologist Jeff Ray visited H.D. Staples Elementary School in Joshua as part of "Weather on Wheels."

Voters headed to the polls Saturday for municipal elections.

A Weather Alert has been issued for the overnight hours as rain and thunderstorms will move into North Texas from the west.

A storm chaser not only captured a massive tornado touching down in Hawley, Texas on Thursday but ended up rescuing a family of four whose home was destroyed by it.

Dallas artist Roberto Marquez traveled to the Rafah Crossing in Egypt, the U.S. capital and will attend this weekend's statewide protest in Austin.

On Friday, hundreds of thousands of fans gathered outside and all around Globe Life Field in Arlington to celebrate the Texas Rangers historical World Series win!

Babies in the neonatal intensive care unit at several Texas Health hospitals were dressed in creative costumes for Halloween.

Is that the smell of cotton candy, beignets and brisket wafting over Fair Park? It sure is, and we are here for it!