CBS News Live
CBS News Texas: Local News, Weather & More
Watch CBS News





Arlington Police are still looking for a key piece of evidence in the deadly school shooting on Wednesday and many questions remain about what happened.

Police arrested 17-year-old Julian Howard and charged him with one count of murder in the death of Etavion Barnes, 18.

The 2024 NFL Draft kicks off Thursday night in Detroit.

Saint Andrews United Methodist Church is opening its door to neighbors who may need a different space to sort out a difficult day.

The victims were taken to a local hospital and are in stable condition.

Students said this was about showing solidarity and not wanting the university to be associated with Israel's fight.

The shooting occurred at an apartment complex in the 2700 block of Lawrence Road and near NE Green Oaks Boulevard and North Collins Street.

The "all in" comment has been mis-interpreted by some fans and media members, who became critical of Jones for not being "all in" on the free agent market this spring.

There may finally be a resolution after a Studio 6 Motel was planned to start construction across the street from Basswood Elementary School back in February.

"If we were to lose control of the Corporation and its assets it would allow the Defendants to remove us from our home, as they have already threatened to do," the Rev. Mother wrote. "I pray they be stopped."

Don Steven McDougal, a family friend, was indicted by a Polk County grand jury in connection with the death of an 11-year-old girl.

Bois D'Arc Lake is now open to families, boaters and fishers just an hour northeast of DFW in Fannin County.

The Cowboys originally had the number 24 pick for the first round, but traded back in the draft and moved to 29.

Two Roosevelt High School of Innovation students were shot near the Cedar Crest Golf Course in Dallas on Thursday.
A Plano massage parlor that police say was engaging in prostitution has shut down its business.

The sheriff said he is asking for more resources for inmates dealing with mental health challenges.
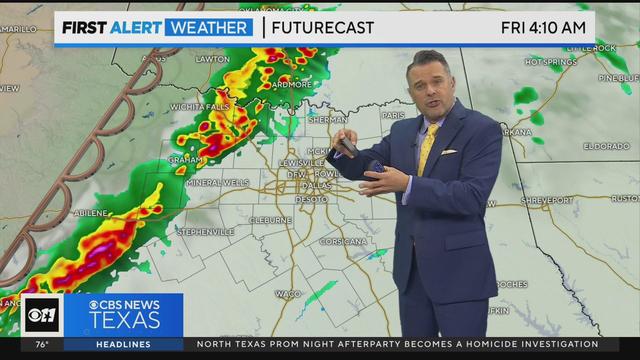
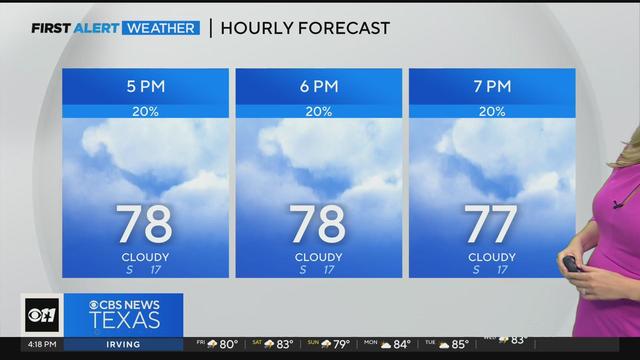
Rainy weekend ahead after first round hits North Texas on Friday morning.

Early morning and daytime storm threats expected Friday in North Texas.

The First Alert Weather Team issued Weather Alerts for Friday, Saturday, and Sunday.
A few strong to severe storms are possible overnight into Friday.

A scammer a North Texas woman met on Instagram claimed to be a German cardiologist, and for months, the two messaged back and forth, building what she thought was a true relationship.

Texas police departments have the discretion to determine the frequency and extent of additional driving training for their officers. While some require driving training yearly or every other year, others do not.

Some departments opt to melt the firearms down, while others choose to crush them. However, there are instances where firearms, or at least parts of them, escape destruction altogether.

Several police departments told the CBS News Texas I-Team they were unaware of this practice, even though it was stated in the contracts they signed with the company, Gulf Coast GunBusters.

It's a complicated process that not everyone qualifies for.

The 2024 NFL Draft kicks off Thursday night in Detroit.

Straight out of the 2024 Big Green NFL Draft Scouting Notebook, here's who the Cowboys won't draft this year, but should.

Noah Hanifin broke a tie with an unassisted goal late in the second period and the Stanley Cup champion Vegas Golden Knights beat the top-seeded Dallas Stars 3-1 on Wednesday night to take a 2-0 lead in the first-round series.

The "all in" comment has been mis-interpreted by some fans and media members, who became critical of Jones for not being "all in" on the free agent market this spring.

We've spent long hours filling up our Big Green NFL Draft Scouting Notebook searching for the perfect draft prospects that can propel the Dallas Cowboys to greener pastures.

Arlington Police are still looking for a key piece of evidence in the deadly school shooting on Wednesday and many questions remain about what happened.

Police arrested 17-year-old Julian Howard and charged him with one count of murder in the death of Etavion Barnes, 18.

The 2024 NFL Draft kicks off Thursday night in Detroit.

Saint Andrews United Methodist Church is opening its door to neighbors who may need a different space to sort out a difficult day.

The victims were taken to a local hospital and are in stable condition.

A scammer a North Texas woman met on Instagram claimed to be a German cardiologist, and for months, the two messaged back and forth, building what she thought was a true relationship.

They found him guilty – now four jurors are explaining how they were convinced to convict Dr. Raynaldo Ortiz.

Texas police departments have the discretion to determine the frequency and extent of additional driving training for their officers. While some require driving training yearly or every other year, others do not.

Some departments opt to melt the firearms down, while others choose to crush them. However, there are instances where firearms, or at least parts of them, escape destruction altogether.

Several police departments told the CBS News Texas I-Team they were unaware of this practice, even though it was stated in the contracts they signed with the company, Gulf Coast GunBusters.

The Supreme Court convened to consider whether former President Donald Trump is entitled to broad immunity from criminal charges in the 2020 election case.

A Texas grand jury indicted more than 140 migrants on misdemeanor rioting charges over an alleged mass attempt to breach the U.S.-Mexico border, a day after a judge threw out the cases.

Regulators prohibit new noncompetes, which impede millions of U.S. workers from getting a better job.

Starting in September of 2024, the year-long residency will allow new teachers to work with veteran teachers before taking on their own classroom.

President Biden signed a foreign aid package into law that includes a potential ban on TikTok in the U.S. Here's what experts say could happen next.

Self-driving 18-wheelers have longtime truckers worried about their livelihood and others concerned that the technology needs more testing to make sure the public is safe.

McDonald's concept restaurant CosMc's has taken its drink-focused menu to Dallas for its second-ever location.

With the country on the cusp of greeting the return of spring, a warm-weather treat is once again available for free for a limited time only.

Kelli and Michael Regan were looking for a new dog. The breeder they found online asked them to pay with gift cards.

Target, looking for ways to add sales, is relaunching its Target Circle loyalty program including a new paid membership with unlimited free same-day delivery in as little as an hour for orders over $35.

The CDC estimates the U.S. could reach 300 measles cases in 2024 — more than the recent peak two years ago.

Organic option is best when buying certain produce, especially blueberries, nonprofit group says in analysis of chemical residues.

The $872 million most likely excludes any amount UnitedHealth may have paid to hackers in ransom.
More than 20 people have been stricken after getting fake or mishandled injections in homes and spas, feds warn.

George Schappell and sister Lori, of Reading, Pa., were the world's oldest conjoined twins, according to the Guinness Book of World Records.

Within hours of the vote, the U.S. Chamber of Commerce announced it would sue to block the ban. Dallas employment attorney Rogge Dunn predicts employees will ultimately win this battle.

The closure affects both Dom's locations in Chicago, and all 33 Foxtrot stores in Chicago, Texas, and the Washington D.C. area.

Texas law SB 14 prohibits drug and surgical "gender transition" interventions for minors.

The projects are expected to create at least 17,000 construction jobs and 4,500 manufacturing jobs.

After more than 40 years in business, 99 Cents Only Stores, a discount chain, announced on Thursday that it will close all 371 of its locations and cease operations.

The 2024 NFL Draft kicks off Thursday night in Detroit.

Straight out of the 2024 Big Green NFL Draft Scouting Notebook, here's who the Cowboys won't draft this year, but should.

Noah Hanifin broke a tie with an unassisted goal late in the second period and the Stanley Cup champion Vegas Golden Knights beat the top-seeded Dallas Stars 3-1 on Wednesday night to take a 2-0 lead in the first-round series.

The "all in" comment has been mis-interpreted by some fans and media members, who became critical of Jones for not being "all in" on the free agent market this spring.

We've spent long hours filling up our Big Green NFL Draft Scouting Notebook searching for the perfect draft prospects that can propel the Dallas Cowboys to greener pastures.

Harvey Weinstein's 2020 conviction on felony sex crime charges has been overturned by the State of New York Court of Appeals.

Mary J. Blige, Cher, Foreigner, A Tribe Called Quest, Kool & The Gang, Ozzy Osbourne, Dave Matthews Band and Peter Frampton have been named to the Rock & Roll Hall of Fame.

Taylor Swift broke her own records, Spotify said, and now owns the record for the top three most-streamed albums in a single day.

The singer was found deceased at her home, a representative said.

Anticipation was growing at a fever pitch before Taylor Swift's latest album, "The Tortured Poets Department," dropped at midnight EDT. But it turned out it's actually a double album.

The Cowboys originally had the number 24 pick for the first round, but traded back in the draft and moved to 29.

Two Roosevelt High School of Innovation students were shot near the Cedar Crest Golf Course in Dallas on Thursday.

A Plano massage parlor that police say was engaging in prostitution has shut down its business.

The sheriff said he is asking for more resources for inmates dealing with mental health challenges.

Rainy weekend ahead after first round hits North Texas on Friday morning.

Dallas artist Roberto Marquez traveled to the Rafah Crossing in Egypt, the U.S. capital and will attend this weekend's statewide protest in Austin.

On Friday, hundreds of thousands of fans gathered outside and all around Globe Life Field in Arlington to celebrate the Texas Rangers historical World Series win!

Babies in the neonatal intensive care unit at several Texas Health hospitals were dressed in creative costumes for Halloween.

Is that the smell of cotton candy, beignets and brisket wafting over Fair Park? It sure is, and we are here for it!

No one puts these dolls back in their boxes. Babies in the neonatal intensive care unit at Texas Health Harris Methodist Hospital Southwest Fort Worth are pretty in pink!